Electric vehicles present unique testing challenges
Rob Plumridge, Sciemetric leak application engineer, discusses best practices for leak testing e-vehicle batteries and how the 3520 Series Leak Test can be used for this purpose, with Assembly Magazine.
Test Equipment Spotlight: QualityWorX CTS DataHub
Cincinnati Test Systems and Sciemetric have brought the data management and analytics of Manufacturing 4.0 to leak and flow testing with their first collaboration as part of the TASI Group. Learn more about the QualityWorX CTS DataHub.
5 ways to improve your production line in 2020
The savings of commonality on the production line
There is a tendency to think about the line in terms of individual process, with separate solutions for force-distance monitoring, leak testing, torque testing, etc., rather than as a whole—but it doesn't have to be this way. With one common solution, like the sigPOD, one platform can do it all. Learn how commonality on the production line helps manufacturers save time and money.
Go beyond station checks to improve product quality and line efficiency: The value of process monitoring and data analytics
5 tips to improve your production line in 2019
With each new year, many of us take time to reflect on our past and set goals for the future. Why not apply the same exercise to your production line? To help you improve your production line in 2019, we have provided the following tips, chock full of insights from our experts.
Sciemetric data analytics tools enabled engine manufacturer to link EOL failure to cracked check valves—see how!
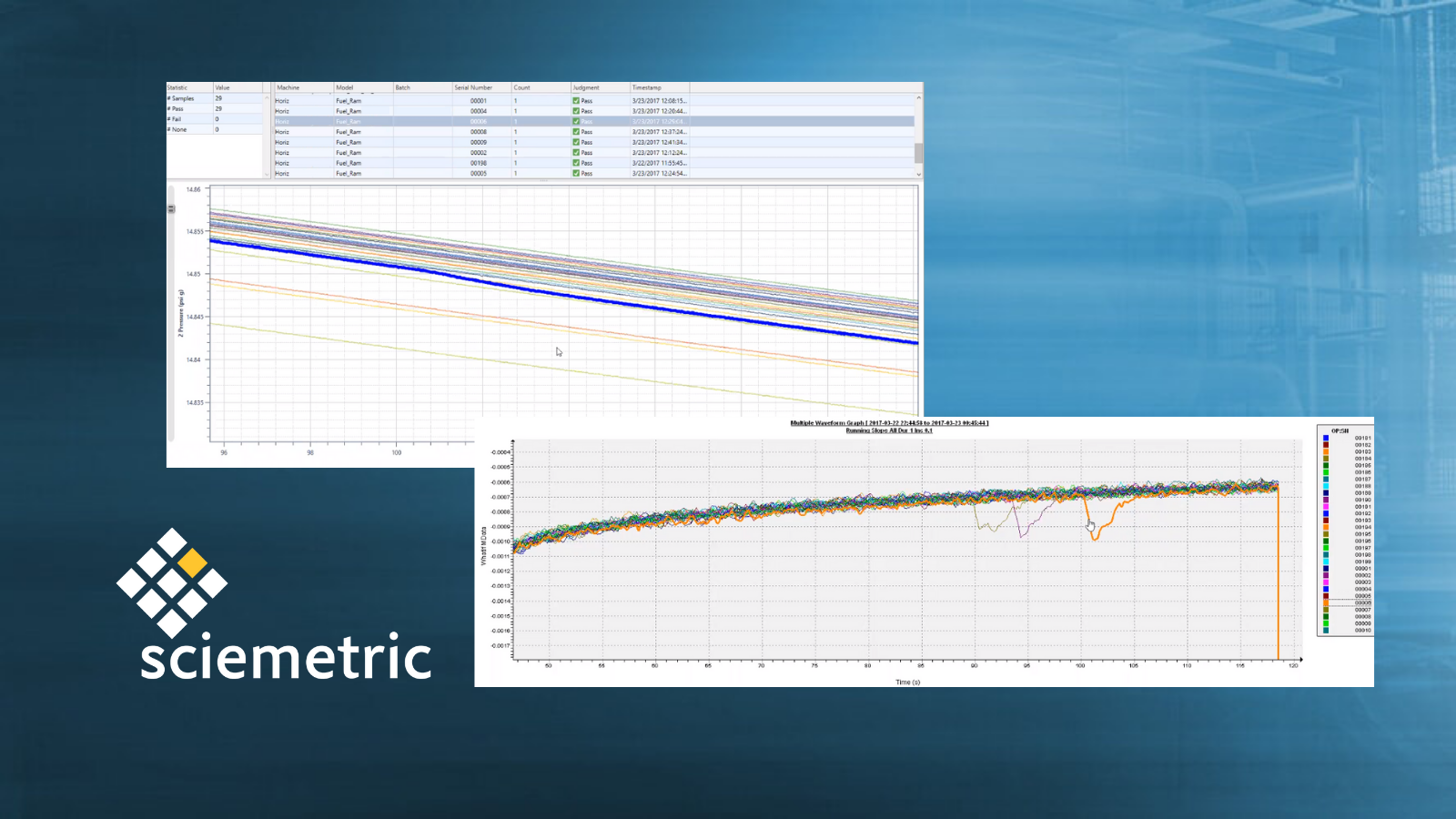
How to catch faulty fuel rail insertion with digital process signature analysis
Tearing down an engine to find a problem when it fails an end-of-line test is costly and time-consuming. It’s much better to identify a quality issue upstream on the production line where it occurs. Learn how an automaker used digital process signatures to adjust their fuel rail insertion parameters to catch faulty insertions before they reach the end of the line.
Turning a regulatory headache into a competitive advantage: Understanding 21 CFR Part 11 and EU Annex 11 compliance
This article by Cincinnati Test Systems' David Kralovetz, market specialist for medical devices, discusses how to balance the implementation of Industry 4.0 monitoring and data analytics while abiding by the regulatory requirements laid out by the FDA's Title 21 CFR Part 11 and European Medicines Agency Annex 11. Read the article to learn more.
Signature Analysis Beyond Press Force
Sciemetric CEO Nathan Sheaff discusses with Assembly Magazine how sensor technology has had to evolve to ensure accurate digital process signature analysis for production processes such as leak testing, crimping, and sound and vibration testing.